Revenue
(2019: £141.3m)
Trading Profit
(2019: £20.9m)
Organic Trading Profit Margin %
(2019: 16.0%)
Scapa Healthcare’s growth strategy is focused on acquiring differentiated technologies and solutions that enhance not only our capabilities, but also our intellectual talent including R&D, engineering, project management, regulatory and quality.
Through our acquisitions and technology transfers, we have strengthened our value proposition as a leading outsource partner with a full range of services including a dedicated innovation team; custom formulations and compounding; analytical and microbiology laboratories; sterilisation services; and quality and regulatory expertise. We are well equipped to capitalise on the outsource trend supporting our customers from concept to commercialisation. Our ability to deliver differentiated solutions to market faster gives our customers a sustainable competitive advantage in the marketplace.
(2019: £141.3m)
(2019: £20.9m)
(2019: 16.0%)
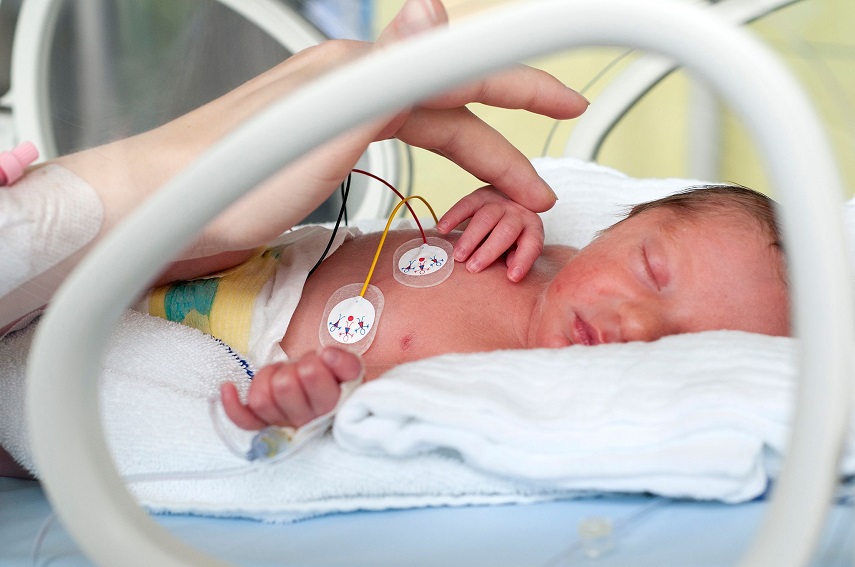
Scapa Healthcare understands the complexity of wound management - from fluid handling to protecting surrounding skin. Our extensive experience co-developing and manufacturing advanced wound dressings and topical skin care solutions allow us to offer unique solutions keeping patient comfort and safety in mind.

Consumer wellness products include first aid and personal care products. Today's consumers are constantly looking for effective solutions to address their personal care needs at home. At Scapa Healthcare, we partner with Consumer Product Companies to identify unmet needs and bring innovative solutions to market rapidly.
Attaching medical devices to the body is particularly challenging as human skin varies from person to person. Scapa Healthcare's MEDIFIX Solutions™ offer a broad portfolio of skin adhesives and medical-grade substrates. We also customise adhesive formulations for unique applications.
Completed consolidation of Inglewood facility into new built-for-purpose Knoxville manufacturing site, allowing for greater operational efficiencies through proximity and scale.
Completed closure of Dunstable facility and transfer of appropriate assets to Gargrave site enhancing Gargrave’s value proposition by offering adhesive coating capabilities to our customers.
Successfully completed transfer of manufacturing equipment and technology for a leading consumer healthcare brand product family into Knoxville facility to support its operational optimisation plan and product innovation.
Secured and completed technology transfer with existing customer to relocate its manufacturing equipment into our Knoxville facility for an additional line of products through a multi-year exclusive manufacturing agreement.
Leveraged our manufacturing, technology and development expertise to enhance our product pipeline across all market segments to support our organic growth strategy and deliver innovative solutions to our customers.
In response to the unprecedented demand for alcohol-based sanitisers during the public health emergency (COVID-19), the FDA released temporary guidance allowing certain entities that are not currently regulated by the FDA as drug manufacturers to prepare and distribute hand sanitiser products for public use.
After carefully reviewing all of our Scapa Healthcare manufacturing sites, it was decided to leverage our current Windsor, Connecticut, facility’s infrastructure for handling alcohol products to temporarily support the production of alcohol-based hand sanitisers. Despite liquid product manufacturing not being Windor’s core competency, Scapa provides an uncompromising focus on quality and regulatory expertise across its operations. The site operations team was therefore able to rapidly register the site with the FDA and obtain the required regulatory licences to start production as quickly as possible.
The team was also able to leverage Scapa Healthcare’s global supply chain to expedite the purchasing of all necessary raw materials. The full operation was up and running in a matter of weeks.Scapa Healthcare continues to look for creative ways to leverage our capabilities to support our global healthcare partners with urgently needed medical products during this global healthcare crisis.

© 2026 Scapa Group Ltd | Legal and Privacy Policy